Maharashtra Board Class 8 Science Solutions Chapter 5 Inside the Atom
Balbharti Maharashtra State Board Class 8 Science Solutions Chapter 5 Inside the Atom Questions and Answers.
Maharashtra State Board Class 8 Science Solutions Chapter 5 Inside the Atom
Class 8 Science Chapter 5 Inside the Atom Textbook Questions and Answers
Class 8 Science Lesson 5 Inside the Atom answers.
Exercise
1. Answer the following.
a. What is the difference in the atomic models of Thomson and Rutherford?
| Thomson Theory | Rutherford Theory |
| 1. The negatively charged electron are embedded in a positively charged solid material which has a spherical shape. | An atom consists of a positively charged nucleus around which the electrons revolve in an orbit. |
| 2. It does not give any details about the atomic nucleus | It mentions that there is a positively charged nucleus at the centre. |
| 3. Electrons are distributed uniformly in an atom. | Electrons revolve around the nucleus. |
| 4. An atom is spherical in shape | An atom has a solid core at the centre defined as the nucleus which is surrounded by the electrons |
| 5. It does not mention the constituents of nucleus | This theory clearly mentions that nucleus is a composition of neutrons and protons |
b. What is meant by valency of an element? What is the relationship between the number of valence electrons and valency?
Ans:
Valency means the number of chemical bonds formed by an atom.
Valency of an atom is determined by the configuration of its outermost shell. Therefore the outermost shell is called valence shell and the electrons in the outermost shell are called valence electrons.
For example:
Valancy:
The electronic configuration of Sodium (Na) is 11 i.e. 2, 8, 1. Hence, the valency of Na is 1.
The electronic configuration of Chlorine (Cl) is 17 i.e. 2, 8, 7. Hence, its valency is 1.
Number of valence electrons:
The electronic configuration of Sodium (Na) is 11 i.e. 2, 8, 1. Hence, the number of valence electrons is 1.
The electronic configuration of Chlorine (Cl) is 17 i.e. 2, 8, 7. Hence, its number of valence electrons is 7.
Thus we see that valency of an element is directly proportional to the number of valence electrons in that atom.
c. What is meant by atomic mass number? Explain how the atomic number and mass number of carbon are 6 and 12 respectively.
Ans: The total number of protons and neutrons in the nucleus of the atom is called atomic mass number. The atomic number that is the proton number of carbon is 6 and the mass number is the total number of protons and neutrons in the carbon that is 6 protons+6 neutrons= 12.
d. What is meant by subatomic particles? Give brief information of three subatomic particles with reference to electrical charge, mass and location.
Ans: The functional and structural unit of matters are the subatomic particles. An atom is formed from the nucleus and the extranuclear part. Thus the nucleus contains subatomic particles such as protons and neutrons in it.
Proton (p): Proton is a positively charged subatomic particle in the atomic nucleus. The positive charge on the nucleus is due to the proton in it. A proton is represented by the symbol ‘p’. Each proton carries a positive charge of +1e. (1e = 1.6 ´ 10-19 coulomb). Mass of one proton is approximately 1u (1 Dalton).
Neutron (n): Neutron is an electrically neutral subatomic particle and is denoted by the symbol ‘n’. The number of neutron in the nucleus is denoted by the symbol ‘N’. Atomic nuclei of all the elements except hydrogen with atomic mass 1u, contain neutrons. The mass of a neutron is approximately 1 u, which is almost equal to that of a proton.
Extranuclear part: The extranuclear part in the atomic structure includes electrons revolving around the nucleus and the empty space in between the nucleus and the electron.
Electron (e- ) Electron is a negatived charged subatomic particle and is denoted by the symbol ‘e- ’. Each electron carries one unit of negative charge (-1e). Mass of an electron is 1800 times less than that of a hydrogen atom. Therefore the mass of an electron can be treated as negligible.
2. Give scientific reasons
a. All the mass of an atom is concentrated in the nucleus.
Ans: The nucleus of the atom contains protons and neutrons. The electrons revolve around the nucleus. The mass of an electron is negligible compared to that of a proton to a neutron. Therefore, the mass of an atom is concentrated in the nucleus.
b. Atoms are electrically neutral.
Ans: An atom is made of two parts that are the nucleus and the extranuclear part. Due to the protons, the nucleus is positively charged. The extranuclear part of an atom is made of negatively charged electrons.
The number of electron in the extranuclear part is equal to the number of proton in the nucleus. Therefore electrical charges are balanced and the atom is electrically neutral.
c. Atomic mass number is a whole number.
Ans: The total number of protons and neutrons in the nucleus of the atom is called atomic mass number. Therefore, the atomic mass number is a whole number.
d. Atoms are stable though negatively charged electrons are revolving within it.
Ans: The entire mass of an atom is concentrated in the nucleus and the positively charged nucleus at the center of an atom. The negatively charged electrons revolve around the nucleus. Electrical charges are balanced and the atoms are stable.
3. Define the following terms
a. Atom:
An atom is the smallest particle of an element which retains its chemical identity in all the physical and chemical changes.
b. Isotope:
Some elements in nature have atoms with the same atomic number but different mass number. Such atoms of the same element having different mass numbers are called isotopes.
c. Atomic number:
The number of electrons or protons in an atom is called the atomic number. It is denoted by Z
d. Atomic mass number:
The total number of protons and neutrons in the nucleus of the atom is called atomic mass number. It is denoted by A.
e. Moderator in nuclear reactor:
Graphite or heavy water is used as moderator for reducing the speed of neutrons.
4. Draw a neat labelled diagram.
a. Ruthrford’s scattering experiment: Refer to textbook pg.no 30 Fig 5.4
b. Thomson’s atomic model: Refer to textbook pg.no 29 Fig 5.3
c. Diagramatic sketch of electronic configuration of Magnesium (Atomic number 12):
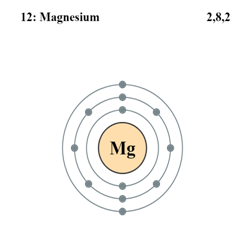
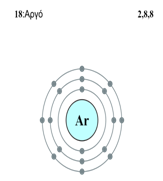
d. Diagramatic sketch of electronic configuration of Argon (Atomic number 18):
5. Fill in the blanks.
a. Electrons, protons, neutrons are the types of subatomic particles in an atom.
b. An electron carries a negative charge.
c. The electron K shell is nearest to the nucleus.
d. The electronic configuration of magnesium is 2, 8, 2. From this it is understood that the valence shell of Magnesium is M.
e. The valency of hydrogen is ‘one’ as per the molecular formula H₂O. Therefore valency of ‘Fe’ turns out to be 3 as per the formula Fe₂O₃.
6. Match the pairs.
Group ‘A’ Group ‘B’
- Proton 3. Positively charged
- Electron 2. Negatively charged
- Neutron 1. Neutral
7. Deduce from the datum provided.
| Datum | To deduce |
| ₁₁²³Na | Neutron number |
| ¹⁴₆C | Mass number |
| ³⁷₁₇Cl | Proton number |
Ans: 1) Neutron number= Mass number-number of protons
= 23-11
= 12
2) Mass number= 14
3) Proton number= Atomic number
= 17
Project :
Explain the atomic models using the material such as old C.D., balloon, thread, marbles, etc .
 JK Academy
JK Academy